Global Lipid Nanoparticle Raw Materials Market Growth Led by North America and Merck KGaA Innovation Leadership
Global Lipid Nanoparticle Raw Materials Market Set to Hit USD 3.44 Billion by 2036 as mRNA Pipelines Shift from Infectious Disease to Oncology
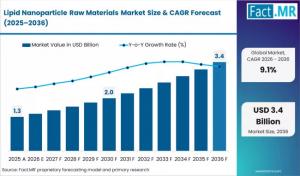
ROCKVILLE, MD, UNITED STATES, April 7, 2026 /EINPresswire.com/ -- The global Lipid Nanoparticle (LNP) Raw Materials Market is entering a high-growth phase, valued at USD 1.44 billion in 2026 and projected to surge to USD 3.44 billion by 2036, according to a comprehensive industry analysis. This represents a robust 9.1% CAGR, driven by an "incremental opportunity" of USD 2.00 billion as drug developers transition from emergency pandemic responses to diversified, long-term therapeutic pipelines in oncology and gene editing.For Details Deep insights, Please Request A sample report for Free: https://www.factmr.com/connectus/sample?flag=S&rep_id=11748
Market Dynamics: From Crisis Response to Clinical Maturity
The supply chain bottleneck for genetic medicines has migrated from mRNA synthesis to the qualification of high-purity lipid raw materials. As clinical-stage pipelines expand, the industry is shifting from ad-hoc, project-based sourcing to formalized, long-term supply agreements.
Key Growth Drivers:
Therapeutic Diversification: Expansion beyond COVID-19 into oncology (37.9% market share), rare diseases, and protein replacement therapies.
Regulatory Scrutiny: Increased demand for GMP-qualified materials as candidates move into Phase II and III trials.
Strategic Stockpiling: Government pandemic preparedness programs in the U.S., EU, and Japan are driving bulk procurement of essential encapsulation components.
Segmental Analysis & Leading Indicators
Metric
2026 Projection
2036 Forecast
CAGR (2026-2036)
Market Value
USD 1.44 Billion
USD 3.44 Billion
9.1%
Dominant Segment
Therapeutics (61.4%)
Therapeutics (Increasing)
—
Lead Product
Kits (54.5%)
Kits / Proprietary Lipids
—
Top Indication
Cancer (37.9%)
Cancer / Infectious Disease
—
By Application:Therapeutics hold a commanding 61.4% share, reflecting the commercialization of LNP-based drugs. However, the Research segment remains vital, capturing 38.6% of the market as academic-industry collaborations explore next-generation delivery vehicles.
By Product:Kits lead the market (54.5%) due to their role in preclinical screening, offering researchers standardized, pre-formulated lipid sets that ensure reproducibility.
Regional Outlook: China and USA at the Forefront
The competitive landscape is bifurcated between established innovation hubs and rapidly scaling manufacturing centers.
China (10.0% CAGR): The fastest-growing market globally, propelled by domestic mRNA vaccine mandates and massive R&D subsidies in biotech clusters like Shanghai and Shenzhen.
United States (8.5% CAGR): Maintains the largest volume of clinical-stage LNP candidates, anchored by the world’s most mature biotechnology infrastructure in Boston and San Francisco.
Europe (8.9% CAGR): Expected to reach USD 890 million by 2036, with Germany (30.2% regional share) leading in manufacturing excellence and the UK emerging as a hub for nanotechnology IP.
Competitive Landscape & Supply Chain Strategy
The market is characterized by high IP barriers, particularly regarding ionizable lipid synthesis. Pharmaceutical procurement teams are increasingly prioritizing dual-sourcing strategies to mitigate the risks associated with single-source dependency during late-stage clinical trials.
Key Industry Participants Include:
Merck KGaA
Avanti Polar Lipids (Croda International)
CordenPharma International
NOF America Corporation
Evonik Industries
Creative Biolabs
Polysciences Inc.
Analyst Perspective
"The industry is at a critical inflection point," says Shambhu Nath Jha, Principal Consultant at Fact.MR. "Drug developers advancing LNP candidates face supplier audit timelines that can extend clinical schedules by months. Success in this market is no longer just about the mRNA cargo; it is about securing a compliant, scalable supply of the lipid matrix that carries it."
Executive Takeaways for Decision-Makers
Investment Opportunity: The USD 2.00 billion incremental growth is concentrated in GMP-grade ionizable lipids and specialized formulation services.
Risk Mitigation: Developers must initiate dual-source qualification during the preclinical phase to avoid "qualification bottlenecks" during Phase III.
Future Outlook: Next-generation LNPs focusing on extra-hepatic targeting (reaching organs beyond the liver) will be the next major frontier for raw material innovation.
For instant access to this report, click “Buy Now” or connect with our analyst for customization: https://www.factmr.com/checkout/11748
To View Our Related Report:
Phospholipids Market: https://www.factmr.com/report/phospholipids-market
Mannosylerythritol Lipid (MEL) Market: https://www.factmr.com/report/mannosylerythritol-lipid-market
Toughening Nanoparticle Additives Market: https://www.factmr.com/report/toughening-nanoparticle-additives-market
Zinc Oxide Nanoparticle Market : https://www.factmr.com/report/4790/zinc-oxide-nanoparticles-market
S. N. Jha
Fact.MR
+1 628-251-1583
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.